- Blog
- Gmt 8 time zone
- Automatic macro meal planner
- Printable a6 expense tracker
- Online apostolic pentecostal bible college
- Dmv title duplicate form
- Fox 5 dc news anchor fired
- Soccer position numbers 4 4 2
- Command to give invisible item frame
- Keyshot 8 download with crack
- Cyberghost chrome review
- Synthesia download free full version mac
- Template commercial invoice
- Decibel relative scale in negative
- Noteburner spotify music converter erfahrungen
- Remote play playstation 3 pc download
- Allura script free font download
- Monitor temps on pc
- Speed test my internet connection
- Pubg mobile hack for pc emulator
- Minecraft 1-12-2 wurst hack client
- Sketchpad 5-0 sketchpad 5-0 online
- Download keygen x force autocad 2014 bagas31
- Pantone mood board template
- Cheat engine hacks for minecraft bedrock edition
- Cacl molar mass periodic table
- Are breakaway cat collars necessary
- Structural functionalism theory vs conflict theory
- Wheel of life template pdf
- Bloodborne emulator pc
- Pokemon omega ruby rom download citra
- Snipping tool free download windows 10
- Og youtube downloader pc windows 7
- Density water kgl
- Auto clicker murgee free download
- Bagpipes vst free download
- Keno numbers ky
- Rfactor 2 original car and track list
- Sailboat retrospective generator
- Dsm 5 diagnostic criteria for ptsd pdf
- Best free vst plugins for reaper
- Apple pages resume templates 2020
- Home depot animal screen
- Among us svg cricut
(1 u is equal to 1/12 the mass of one atom of carbon-12)
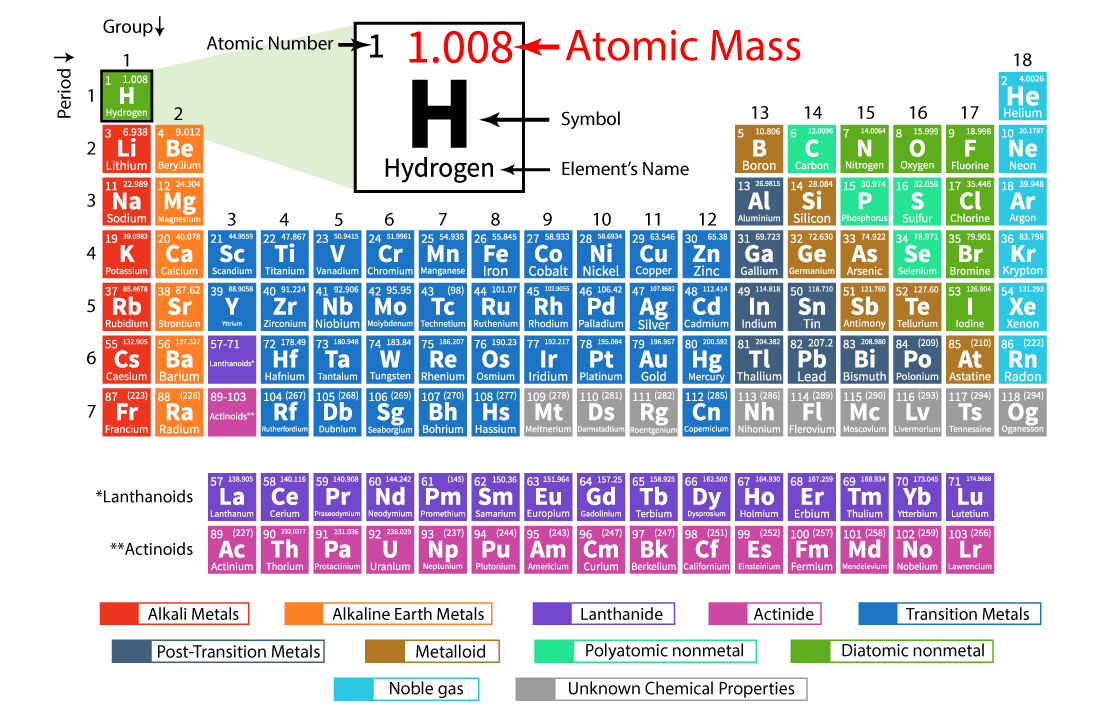
CO 2 has one carbon atom and two oxygen atoms.Oxygen (O) has an atomic mass of about 16.00 amu.
Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find atomic masses: look up the atomic masses of each element present in the compound.For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound.One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) Mole is a standard scientific unit for measuring large quantities of very small entities such as atoms and molecules.Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol.Calcium carbonate appears as a white, odorless powder or solid with a crystalline structure.ĬaCO 3 + H 3PO 4 = Ca 3(PO 4) 2 + H 2CO 3Ĭomputing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'.
- Blog
- Gmt 8 time zone
- Automatic macro meal planner
- Printable a6 expense tracker
- Online apostolic pentecostal bible college
- Dmv title duplicate form
- Fox 5 dc news anchor fired
- Soccer position numbers 4 4 2
- Command to give invisible item frame
- Keyshot 8 download with crack
- Cyberghost chrome review
- Synthesia download free full version mac
- Template commercial invoice
- Decibel relative scale in negative
- Noteburner spotify music converter erfahrungen
- Remote play playstation 3 pc download
- Allura script free font download
- Monitor temps on pc
- Speed test my internet connection
- Pubg mobile hack for pc emulator
- Minecraft 1-12-2 wurst hack client
- Sketchpad 5-0 sketchpad 5-0 online
- Download keygen x force autocad 2014 bagas31
- Pantone mood board template
- Cheat engine hacks for minecraft bedrock edition
- Cacl molar mass periodic table
- Are breakaway cat collars necessary
- Structural functionalism theory vs conflict theory
- Wheel of life template pdf
- Bloodborne emulator pc
- Pokemon omega ruby rom download citra
- Snipping tool free download windows 10
- Og youtube downloader pc windows 7
- Density water kgl
- Auto clicker murgee free download
- Bagpipes vst free download
- Keno numbers ky
- Rfactor 2 original car and track list
- Sailboat retrospective generator
- Dsm 5 diagnostic criteria for ptsd pdf
- Best free vst plugins for reaper
- Apple pages resume templates 2020
- Home depot animal screen
- Among us svg cricut
